 
SpaceNext50 Britannica presents SpaceNext50, From the race to the Moon to space stewardship, we explore a wide range of subjects that feed our curiosity about space!.Learn about the major environmental problems facing our planet and what can be done about them! Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century.Britannica Beyond We’ve created a new place where questions are at the center of learning.100 Women Britannica celebrates the centennial of the Nineteenth Amendment, highlighting suffragists and history-making politicians.COVID-19 Portal While this global health crisis continues to evolve, it can be useful to look to past pandemics to better understand how to respond today.Student Portal Britannica is the ultimate student resource for key school subjects like history, government, literature, and more.Demystified Videos In Demystified, Britannica has all the answers to your burning questions.This Time in History In these videos, find out what happened this month (or any month!) in history.#WTFact Videos In #WTFact Britannica shares some of the most bizarre facts we can find.Britannica Classics Check out these retro videos from Encyclopedia Britannica’s archives.Britannica Explains In these videos, Britannica explains a variety of topics and answers frequently asked questions.Protium is made up of one proton and no neutrons. The letter H is used to denote protium, which is the normal form of hydrogen. An element’s isotope is described as an atom with the same number of protons but a different number of neutrons. The basic hydrogen atom, protium, has a single proton surrounded by a single electron. Their chemical properties should be similar because the isotopes have the same atomic number and electronic configuration. They do, however, differ in terms of the number of neutrons. The outer shell of all three hydrogen isotopes has one electron while the nucleus has only one proton. Due to the enormous differences in mass, they have different physical properties. However, because of the varied bond disassociation enthalpies, they have distinct reaction rates. Isotopes have similar chemical properties because their electrical arrangement is the same. 
Only tritium, out of these three hydrogen isotopes, is radioactive in nature and generates low-energy β-particles. Protium is the most common form of hydrogen, with deuterium accounting for 0.0156% of all hydrogen on the planet’s surface. There are no neutrons in protium, while one neutron is present in deuterium and two neutrons are present in tritium. Protium atoms make up the majority of hydrogen found in nature. 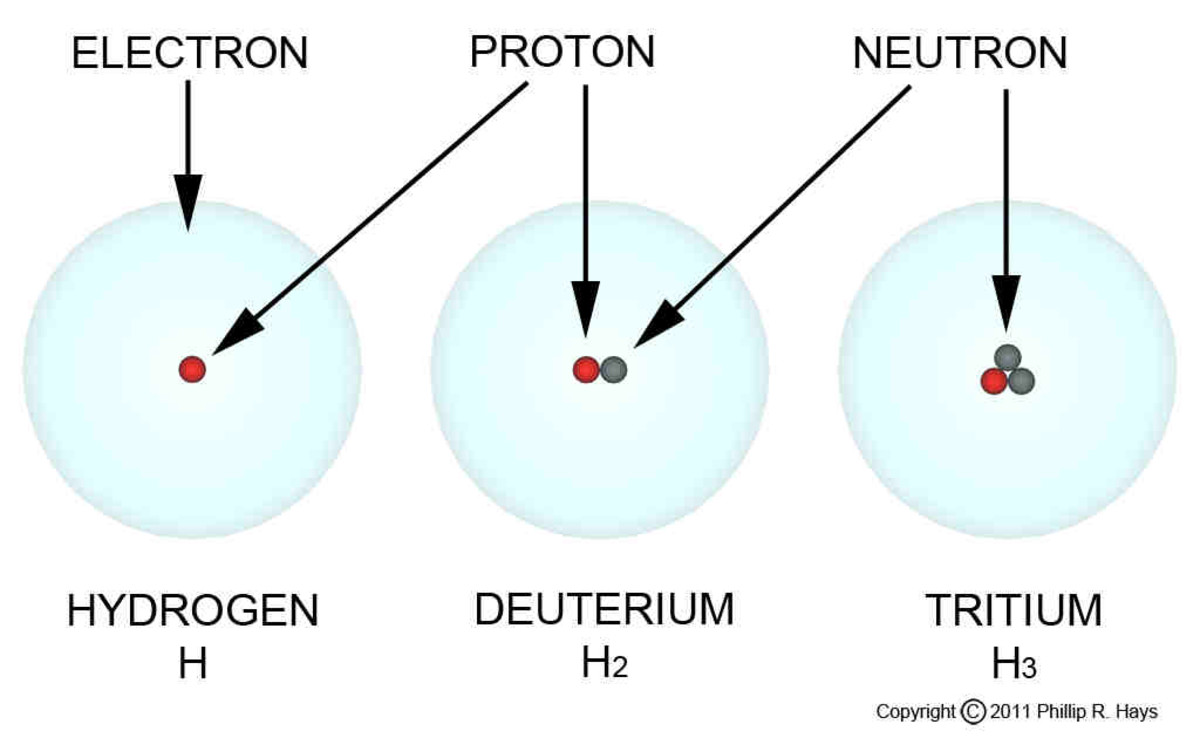
In nature, the relative abundances of three isotopes are 1: 1.56×10 -2: 1×10 -18, respectively. Protium, deuterium, and tritium are the three naturally occurring hydrogen isotopes.ĭue to the various numbers of neutrons in each isotope, the isotopes differ. Hydrogen has three isotopes, each with a mass number of 1, 2, or 3. Isotopes are distinct forms of the same element with different mass numbers but the same atomic number. Hydrogen atoms make up about 15% of all the atoms on the earth. It is tenth in the order of abundance in crystal rocks. It’s not found in our atmosphere because the earth’s gravitational pull isn’t strong enough to hold light hydrogen molecules. It makes up 15.4% of the earth’s crust and seas. Hydrogen is found in a variety of minerals, including coal, petroleum, oil, and natural gas. Hydrogen is found on Earth in a variety of forms, mostly in combination with oxygen in the form of water, and in combination with carbon, nitrogen, and halogens in the form of organic matter in plant and animal tissues, carbohydrates, proteins, and other substances. ISRO CS Syllabus for Scientist/Engineer Exam.ISRO CS Original Papers and Official Keys.GATE CS Original Papers and Official Keys.DevOps Engineering - Planning to Production.Python Backend Development with Django(Live).Android App Development with Kotlin(Live).Full Stack Development with React & Node JS(Live).Java Programming - Beginner to Advanced.Data Structure & Algorithm-Self Paced(C++/JAVA).Data Structure & Algorithm Classes (Live). 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
